
Endo-Free Plasmid Maxiprep Kit
Endo-Free Plasmid Maxiprep Kit 10T
500-1800 µg of high-purity plasmid DNA can be extracted from 100 mL of bacterial culture.

Endo-Free Plasmid Maxiprep Kit 10T
500-1800 µg of high-purity plasmid DNA can be extracted from 100 mL of bacterial culture.
Product Information
|
Product Name |
Cat. No. |
Spec. |
|
Endo-Free Plasmid Maxiprep Kit |
G3645-10T |
10T |
Product Description/Introduction
This kit is suitable for extracting various endotoxin-free plasmid DNA from cultured bacteria. Adopting the unique silica membrane adsorption technology and matching with endotoxin-removing solutions Buffer ER and Buffer ED, the kit can obtain high-purity plasmid DNA with a concentration of 500-1800 μg while effectively removing endotoxins. The plasmid DNA extracted using this kit can be applied to biological experiments such as digestion reaction, ligation reaction, PCR amplification, sequencing, transformation and transfection of various cells.
Storage and Shipping Conditions
RNase A is shipped with wet ice and stored at -20°C. Other reagents are shipped and stored at room temperature, valid for 12 months.
Product Content
|
Component Number |
Component |
G3645-10T |
|
G3645-1 |
Buffer BL |
30 mL |
|
G3645-2 |
Buffer P1 |
100 mL (Add RNase A before use) |
|
G3645-3 |
Buffer P2 |
100 mL |
|
G3645-4 |
Buffer P3 |
100 mL |
|
G3645-5 |
RNase A |
200 μL |
|
G3645-6 |
Buffer ER |
30 mL |
|
G3645-7 |
Buffer ED |
120 mL |
|
G3645-8 |
Buffer PW |
70 mL (Add 163 mL of anhydrous ethanol before use) |
|
G3645-9 |
Buffer TE |
30 mL |
|
G3645-10 |
Column Filters |
10 pcs |
|
G3645-11 |
HiBind DNA Columns |
10 pcs |
|
G3645-12 |
50 mL Collection Tubes |
10 pcs |
|
Manual |
1 copy |
|
Before starting (please read carefully)
1. Bring your own anhydrous ethanol and 50 mL Nuclease-free centrifuge tubes.
2. Add all the RNase A provided in the kit to Buffer P1 before use, and it can be stored at 2-8°C for 6 months.
3. Please add 163 mL anhydrous ethanol to Buffer PW before first use.
4. If Buffer P2 precipitates, please heat it in a water bath at 37°C for a few minutes to restore clarification. After using Buffer P2, the lid should be closed immediately to avoid long-term contact with air.
5. Please pre-cool Buffer P3 at 4°C before use.
Assay Protocol/Procedures
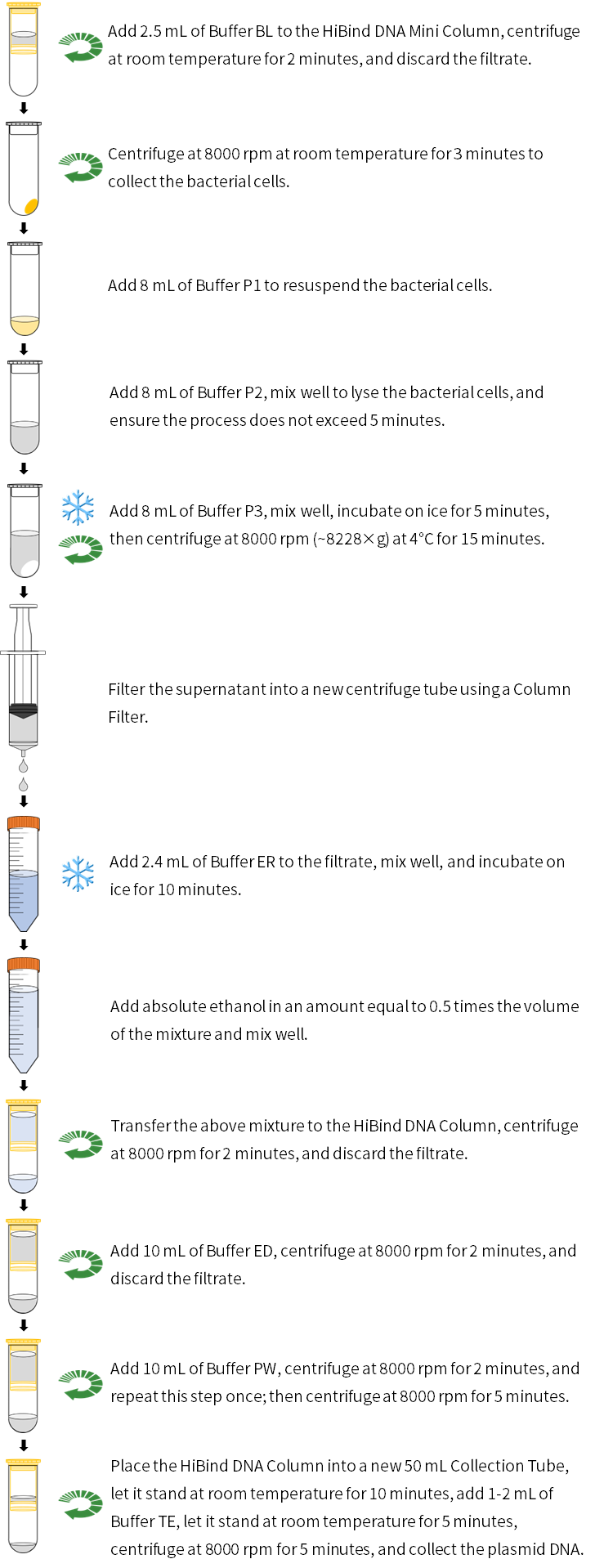
1. Column balance: Add 2.5 mL of Buffer BL to the HiBind DNA Column (place the HiBind DNA Column into a 50 mL Collection Tube in advance), centrifuge at 8000 rpm (~8228×g) at room temperature for 2 minutes, discard the waste liquid, and place the HiBind DNA Column back into the Collection Tube (it is recommended to use the processed column immediately).
2. Take 100 mL of overnight-cultured bacterial solution (for low-copy plasmids, it is recommended to take 200 mL of overnight-cultured bacterial solution), centrifuge at 8000 rpm (~8228×g) at room temperature for 3 minutes to collect the bacterial cells in a 50 mL centrifuge tube (try to remove all the supernatant).
3. Add 8 mL of Buffer P1 (please first check whether RNase A has been added), and use a pipette or vortex oscillator to thoroughly suspend the bacterial cells (it is essential to fully disperse the bacterial cells; otherwise, lysis will be affected, resulting in low quality and purity of the extracted plasmids).
4. Add 8 mL of Buffer P2 and immediately invert gently up and down 8-10 times to fully lyse the bacterial cells (this step must not involve vigorous shaking and should be completed within 5 minutes). At this point, the solution should become clear and viscous. If it does not become clear, it may be due to an excessive amount of bacterial cells; invert several more times until the solution becomes transparent.
5. Add 8 mL of pre-chilled Buffer P3 (pre-cooled at 4°C in advance), immediately invert gently up and down 10-12 times. Compact aggregates will form in the solution. Place on ice for 5 minutes, then centrifuge at 8000 rpm (~8228×g) at 4°C for 15 minutes. Slowly pour all the supernatant into the Column Filter, push the plunger to filter, and collect the filtrate in a nuclease-free 50 mL centrifuge tube (prepared by user).
6. Add 2.4 mL of Buffer ER, invert to mix, then place on ice for 10 minutes (invert to mix 3-5 times during this period). The solution will appear transparent blue.
7. Add anhydrous ethanol equal to 0.5 times the volume of the above solution, invert up and down to mix, then transfer to the HiBind DNA Column (do not add more than 10 mL of liquid each time).
8. Centrifuge at 8000 rpm (~8228×g) at room temperature for 2 minutes, discard the waste liquid in the HiBind DNA Column, and place the HiBind DNA Column back into the Collection Tube (the solution from step 7 can be passed through the column multiple times).
9. Add 10 mL of Buffer ED to the adsorption column, centrifuge at 8000 rpm (~8228×g) at room temperature for 2 minutes, discard the waste liquid in the HiBind DNA Column, and place the HiBind DNA Column back into the Collection Tube.
10. Add 10 mL of Buffer PW to the HiBind DNA Column, centrifuge at 8000 rpm (~8228×g) at room temperature for 2 minutes, discard the waste liquid in the collection tube, and place the HiBind DNA Column back into the Collection Tube.
11. Repeat step 10.
12. Centrifuge at 8000 rpm (~8228×g) at room temperature for 5 minutes.
13. Place the HiBind DNA Column into a new 50 mL Collection Tube, leave the lid open and let it stand at room temperature for 10 minutes. Add 1-2 mL of Buffer TE dropwise to the middle of the adsorption membrane without touching it, let it stand at room temperature for 5 minutes, then centrifuge at 8000 rpm (~8228×g) for 5 minutes. If it is necessary to increase the plasmid recovery efficiency, the obtained solution can be re-added to the HiBind DNA Column, left to stand at room temperature for 5 minutes, and then centrifuged at 8000 rpm (~8228×g) for 5 minutes.
14. Transfer all the eluate from the 50 mL Collection Tube to a nuclease-free 1.5 mL centrifuge tube and store at -20°C.
Note
1. Please read the Product Manual carefully before use.
2. When filtering with the Column Filter, do not add too much precipitate to avoid clogging the filter.
3. When the extracted plasmid DNA is larger than 10 kb, the amount of bacterial cells collected should be increased.
4. Ethanol should be completely volatilized before eluting the plasmid to prevent residual ethanol from affecting downstream experiments.
5. For your safety and health, please wear a lab coat and disposable gloves during operation.
For Research Use Only!
Flowchart
Use collapsible tabs for more detailed information that will help customers make a purchasing decision.
Ex: Shipping and return policies, size guides, and other common questions.